Self-supervised learning with BYOL for anterior cruciate ligament tear detection from knee MRI
When a tiny ligament causes a big clinical problem
Anterior cruciate ligament (ACL) tears are among the most common and clinically significant knee injuries. MRI is the gold-standard non-invasive tool for visualizing soft tissues, yet ACL tear detection still depends heavily on expert interpretation a process that is time-consuming, experience-dependent, and vulnerable to variability across scanners an
d patient populations. As demand grows, the question becomes practical: how do we scale accurate interpretation without scaling human workload?
The challenge: great models need great labels
Deep learning has already shown strong promise in knee MRI analysis, but most high-performing systems rely on large, carefully labeled datasets. In the real world, labels are expensive: they require radiologists, clear protocols, and time. And even when labels exist, models can lose performance when deployed on data from different hospitals or scanners.
That is where self-supervised learning (SSL) becomes a strategic advantage: instead of learning only from labeled examples, SSL can learn meaningful structure from unlabeled MRI scans, which are far more abundant.
The FAST-AI idea
FAST-AI applies a two-stage pipeline that turns unlabeled MRI data into a powerful diagnostic foundation:
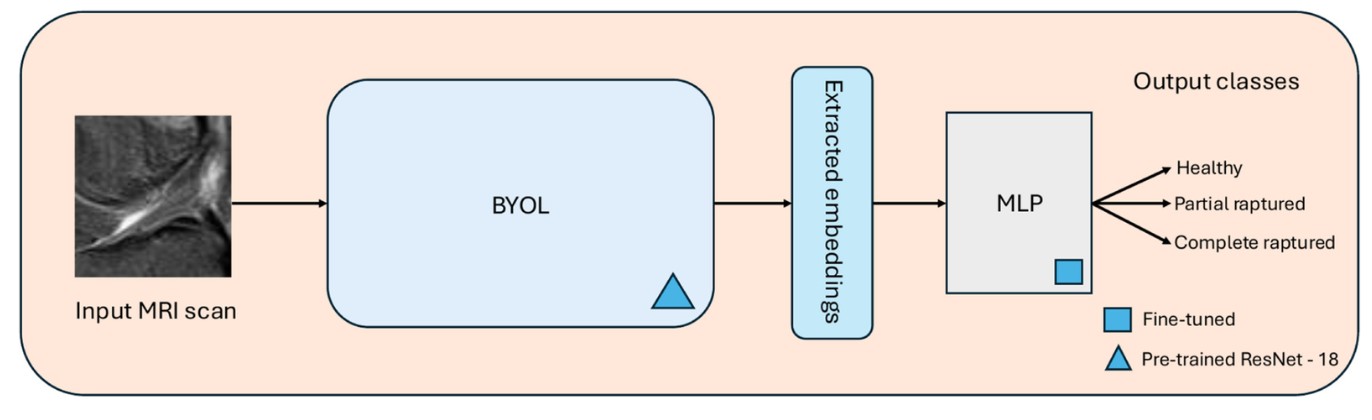
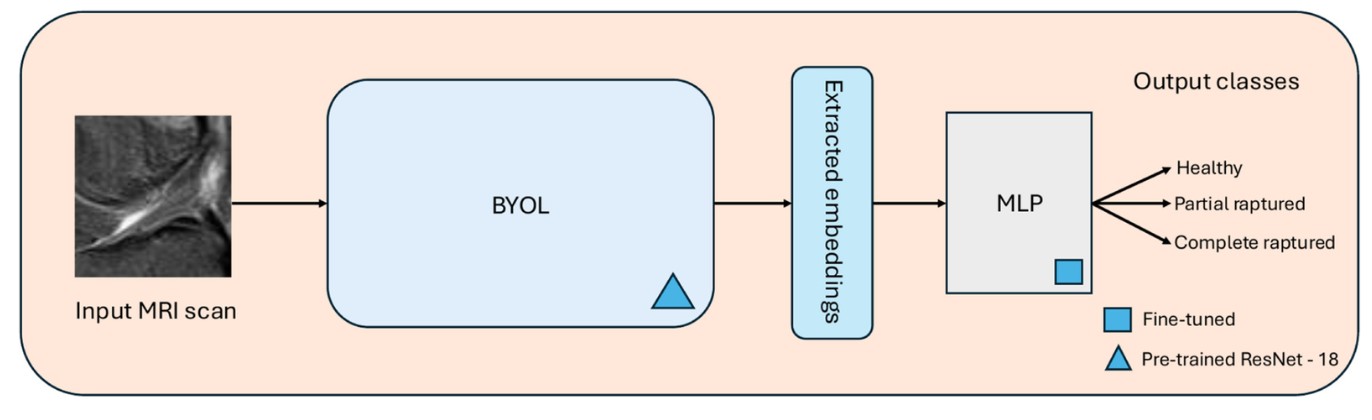
Learn representations without labels (BYOL pretraining)
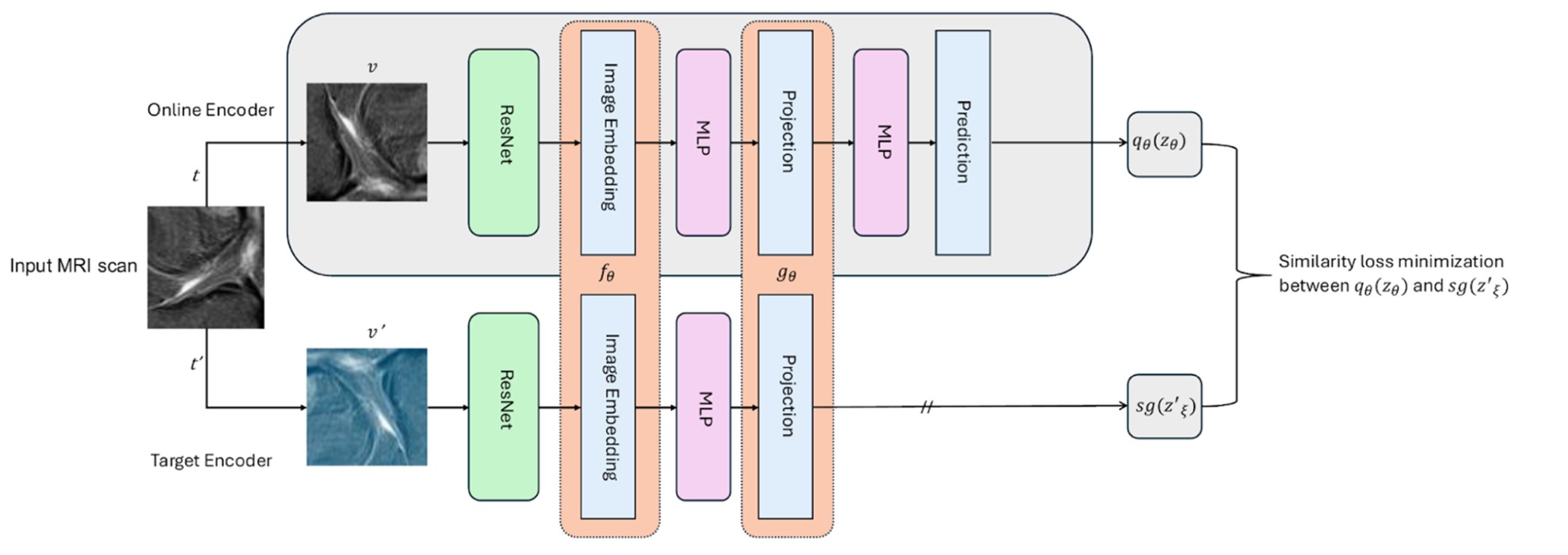
We use Bootstrap Your Own Latent (BYOL) to pretrain a ResNet-18 encoder on unlabeled knee MRIs. BYOL learns robust feature representations by comparing two augmented views of the same image without needing negative samples—which is especially useful in medical imaging where “hard negatives” are not always clear.
Transfer to a clinical task (ACL tear classification)
Once the encoder learns useful MRI features, we extract embeddings and feed them into a lightweight downstream classifier (MLP) to predict ACL status across clinically relevant classes (e.g., healthy vs. tear severity).
In short: unlabeled MRIs teach the model “what matters,” and labeled data teaches it “what to call it.”
Why this matters clinically
The key benefit isn’t only accuracy it’s generalization. SSL-pretrained encoders tend to learn features that transfer better when labeled data is limited, and they can be more resilient across domain shifts (different scanners, protocols, and populations). That’s a real-world requirement for any model that aims to move beyond a single institution.
What we observed
Across model configurations, the strongest results emerged when the BYOL-pretrained encoder was fine-tuned rather than frozen. In our evaluation, the BYOL-pretrained ResNet-18 + MLP (unfrozen encoder) achieved the best overall discrimination performance with an AUC of 0.79 ± 0.05, outperforming ImageNet-pretrained and randomly initialized baselines. Precision/recall trends also improved with fine-tuning, reinforcing a consistent pattern: representation learning helps, but adaptation matters.
Limitations and the next step
This work used a single 2D MRI slice, which may miss important 3D context available across volumes. Future work will expand toward volumetric modeling, explore deeper encoders, and integrate multi-slice or sequence-aware approaches to capture ligament continuity and subtle tear patterns more reliably.
What FAST-AI aims to become
FAST-AI is a step toward clinically useful AI for orthopaedic imaging: a system that learns from what hospitals already have large volumes of unlabeled scans and converts that latent information into decision support for earlier, faster, and more consistent ACL diagnosis.
Funding note: This research was funded by the Ministry of Science and Higher Education of the Republic of Kazakhstan (Grant No. BR24992820).
https://www.sciencedirect.com/science/article/pii/S2215016125005084
